
At this frequency, the only thing that can change the three parameters is temperature. Most commercial microwave systems are set to a frequency of 2450 MHz. 1 Functional groups, temperature, frequency, and volume will all influence the relaxation time of a solvent. The dielectric relaxation time is the time it takes a molecule to achieve 63% of its return to randomized disorder from an organized state after an applied microwave field is removed. Molecular relaxation time has a large effect on these parameters. The three main dielectric parameters, tangent delta, dielectric constant, and dielectric loss, are all related to the ability of a solvent to absorb microwave energy. This will be discussed in greater detail in the following section. It is this value, εʺ, that best provides the organic chemist with the coupling efficiency of a particular solvent. Dielectric loss is the amount of input microwave energy that is lost to the sample by being dissipated as heat. It is defined as the ratio of the dielectric loss, or complexed permittivity ( εʺ), to the dielectric constant ( ε). Tangent delta ( δ), or loss tangent, is the dissipation factor of the sample or how efficiently microwave energy is converted into thermal energy. The ability of a substance to convert electromagnetic energy into heat at a given frequency and temperature is determined by the following equation: tan δ = εʺ/ε. This is because polarization depends on dipole rotation - the ability of a molecule’s dipole to align with a rapidly changing electric field. Molecules with large dipole moments also have large dielectric constants. The magnitude can also be defined as: μ = Qr (μ = dipole moment, Q = charge, and r = distance between charges). One equation used to determine dipole moment is: T = pE (T = torque, p = dipole moment, and E = field strength). It is the product of the distance between the centers of charge in the solvent molecule multiplied by the magnitude of that charge. The dipole moment, which is measured in Debye units (D), is also a mathematical entity. This value, when measured, is dependent on both temperature and frequency. Mathematically, it is the ratio of the electrical capacity of a capacitor filled with the solvent to the electrical capacity of the evacuated capacitor ( ε =C filled /C evacuated). The dielectric constant (ε), also known as the relative permittivity, of a solvent measures its ability to store electric charges. Intrinsically, the dielectric constant, dipole moment, dielectric loss, tangent delta, and dielectric relaxation time all contribute to an individual solvent’s absorbing characteristics. Many factors characterize the polarity of a solvent. In addition, the last section will discuss how to choose a solvent in a microwave-enhanced organic reaction. This chapter will discuss the theory behind solvent polarity and how it pertains to the individual solvents, their physical constants, and how they behave in a microwave field. As discussed in the previous chapter, this interaction leads to a rapid rise in temperature and faster reaction rates. The more polar a reaction mixture is, the greater its ability to couple with the microwave energy. With microwave heating, this becomes a more significant component, as microwaves directly couple with the molecules that are present in the reaction mixture. One of the most important characteristics of a solvent is its polarity. Most reactions take place in solution, and therefore, choice of solvent can be a crucial factor in the outcome of a reaction. 
Solvents play a very important role in organic synthesis. 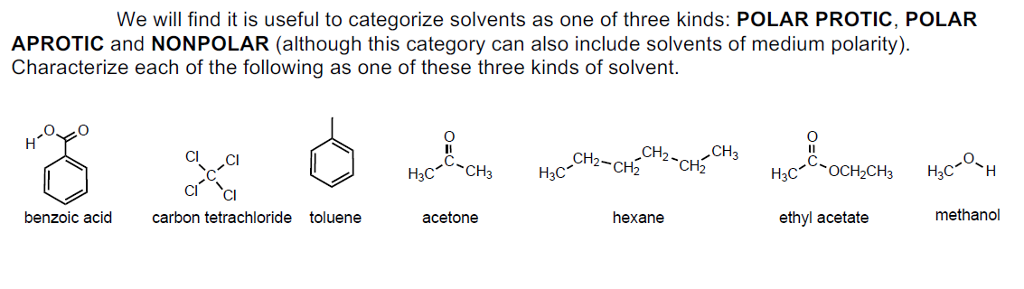
The more efficient a solvent is in coupling with the microwave energy, the faster the temperature of the reaction mixture increases.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
