|
First, HNSCC has a relatively high tumor mutation burden (TMB). The biological rationale for antitumor immunotherapy specifically in HNSCC is built upon several observations. In HNSCC there is a considerable need to improve survival without further exacerbating toxicity.Īntitumor immunotherapy is based upon the principle that adaptations in immune surveillance and the tumor microenvironment allow immune escape. The toxicity of the EXTREME regimen is considerable, with an 82% rate of grade 3–4 adverse events (AE). For recurrent/metastatic (R/M) disease, median survival is only 10.1 months with the historic standard first-line EXTREME regimen using the triplet: cis- or carboplatin, 5-fluorouracil (5-FU) and cetuximab. In addition, the rapid emergence of the human papillomavirus (HPV)-associated subset of HNSCC has motivated novel, immune-based therapies. Despite aggressive multimodal strategies to treat head and neck squamous cell carcinoma (HNSCC) using combinations of surgery, radiotherapy (RT) and chemotherapy, the 5-year overall survival of carcinogen-related HNSCC is only 40–50%. 1 Approximately 430,000 will die from this disease. Worldwide, approximately 830,000 patients develop head and neck cancer each year. In this article we discuss the clinical trials leading to these therapeutic advances and we will review initial results from clinical trials in previously untreated, locally advanced disease, and those using novel combinations of checkpoint inhibitors, co-stimulatory agonists, and therapeutic vaccines. 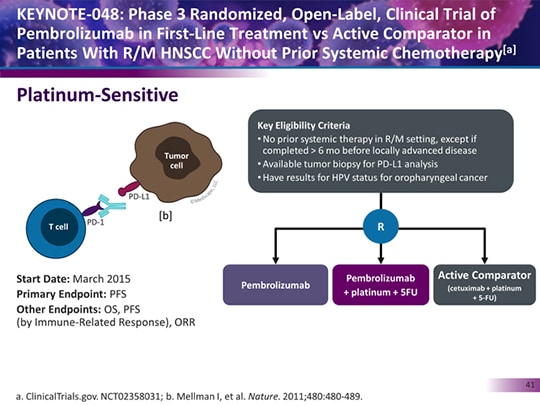
Additionally, for the 85% of patients with PD-L1 CPS ≥1, clinical judgment will guide the choice of pembrolizumab monotherapy or pembrolizumab plus chemotherapy, until more detailed clinical data are forthcoming to better inform this decision. Thus, PD-L1 biomarker testing will be routinely used in R/M HNSCC, and this employs a scoring system that incorporates immune cell staining, referred to as the combined positive score (CPS). Pembrolizumab is now approved as monotherapy in PD-L1 expressing disease (combined positive score ≥1) or in combination with chemotherapy for all patients with R/M HNSCC. Recently, a third phase III randomized trial, KEYNOTE-048, showed that pembrolizumab with chemotherapy was superior to the EXTREME regimen (cis- or carboplatin, 5-fluorouracil (5-FU) and cetuximab) in all patients, and pembrolizumab monotherapy was superior in patients whose tumors express PD-L1 in first-line R/M HNSCC. Two of these phase III randomized trials showed that the anti-PD1 antibodies nivolumab and pembrolizumab were superior to investigators′ choice chemotherapy in second-line platinum-refractory R/M HNSCC. 
Three randomized phase III trials have now conclusively proven that exposure to a PD-1 inhibitor prolongs survival in recurrent/metastatic (R/M) HNSCC, and it is clear that such agents should be used in the management of all patients who do not have contraindications to their use.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
